R&D
Anti-CCL24 Therapeutic Platform
Introduction
Chemomab’s R&D talent, infrastructure and capabilities are paving the way for successful drug introduction from early-stage discovery through clinical development. Our R&D team is investigating the underlying fibrosis biology via various in-vitro and in-vivo models, which are propelling our development of transformative patient therapeutics forward.
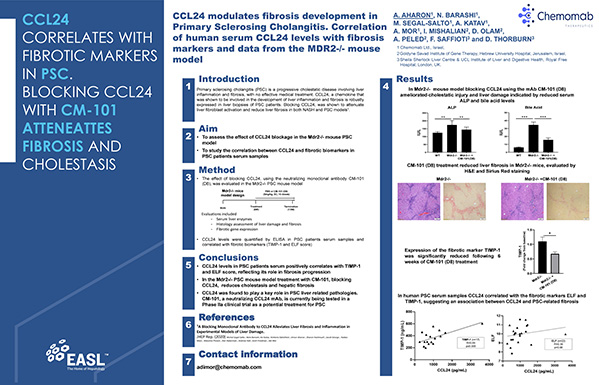
Our science-driven approach explores novel pharmacodynamic parameters from our clinical trials to equip us with powerful biological insights. Using patients’ tissues and serum samples, as well as primary cell cultures, we are developing deeper understanding of CCL24 expression and its specific roles in primary sclerosing cholangaitis (PSC) and systemic sclerosis (SSc) pathologies.
Chemomab leverages collaborations with leading investigators from prestigious research centers around the world to both obtain further evidence on CCL24’s unique role in fibrosis and demonstrate CM-101’s potential therapeutic effects on fibrosis.
CCL24’s Role in the Vicious Cycle of Inflamation and Fibrosis and How CM-101 Neutralizes CCL24, Breaking Vicious Cycle
CCL24’s Role in the Vicious Cycle of Inflamation and Fibrosis and How CM-101 Neutralizes CCL24, Breaking Vicious Cycle
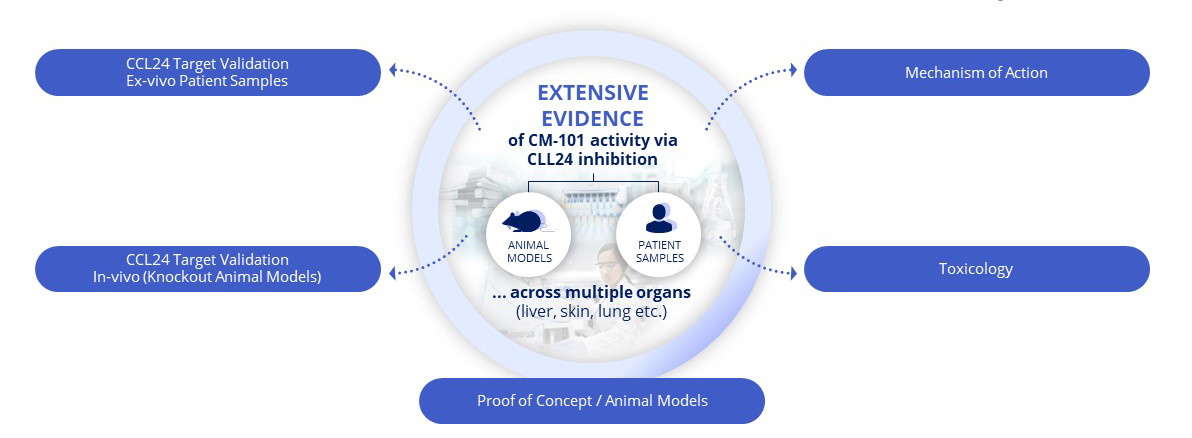
Broad, Diverse Preclinical Evidence De-risks Clinical Development
The Roles of CCL24 and CM-101
About CCL24
Over the last decade, our scientific team discovered the role of CCL24 as a key regulator in fibrotic-inflammatory processes, specifically, the pivotal pro-fibrotic and pro-inflammatory effects of CCL24 in fibrotic diseases.
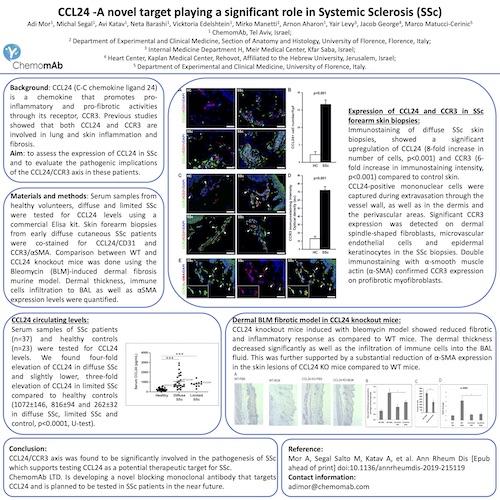
Chemomab observed CCL24 up-regulation and its correlation with disease severity in fibrotic disease tissues, including liver, skin and lung fibrosis related pathologies (e.g. PSC, SSc).
Based on these revelations, Chemomab established a comprehensive preclinical package to support the therapeutic effects of blockading CCL24.
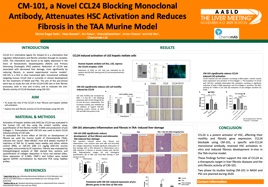
We revealed that CCL24 works through a dual biological pathway directly activating fibroblasts while recruiting immune cells and maintaining the inflammatory environment that is required to enhance fibrosis.
Chemomab proved that inhibiting these pathways by blocking CCL24 prevents and reverses fibrotic tissue growth within the liver, skin and lung.
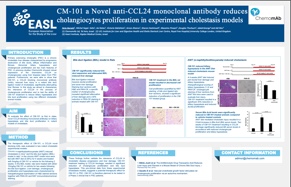
Based on strong human data and relevant disease models, Chemomab developed CM-101, a monoclonal antibody that targets CCL24 and blocks its downstream pathways.
About CM-101 Therapeutic Platform
CM-101 is a novel, safe and potentially effective therapy that fills the huge gap in fibrotic disease care. Treatment with CM-101 revealed strong anti-fibrotic effects, reduced inflammatory injury and significantly improved organ damage. Numerous in-vivo, in-vitro and ex-vivo studies verified that using CM-101 in fibrotic disease models including PSC, SSc, idiopathic pulmonary fibrosis (IPF) and NASH demonstrated potent therapeutic CCL24 blockade effects.
CM-101 is safe and well tolerated up to the highest tested dose in healthy subjects and in fatty liver disease patients with both intravenous and subcutaneous administrations. Both routes of administration support long dosing intervals – once every 2-4 weeks – and provide evidence of target engagement and biological activity in humans.
Based on CM-101’s broad, robust biological effects, favorable tolerability profile and unique mechanism of action, Chemomab believes CM-101 will counteract deleterious pathological processes that drive fibrosis, having the potential to become a leading anti-fibrotic therapeutic agent.
CM-101 phase II trials in patients with PSC and NASH are currently underway.